Asymmetric Ylide MIRC
5 August 2005 Asymmetric cyclopropanation update
So what else is new in the field of asymmetric cyclopropanation. The asymmetric Simmons-Smith reaction has been around since 1992 but the new century has a lot to offer. In one method, part of a larger class of Michael-initiated ring-closure (MIRC) an ylide reacts with a Michael acceptor
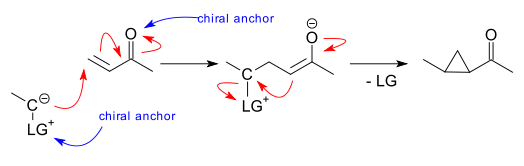
The nucleophilic carbon atom of the ylide reacts with the Michael group first and in the second step the electrophilic part is displaced as the leaving group with ring closure. A chiral moiety can then interact or is part of either with the carbonyl group (amine organocatalysis) or with the leaving group. Pioneering work originates from Johnson & Schroeck using a oxosulfonium methylide (1968 DOI) and from Trost & Hammen (sulfonium ylide, 1973 DOI). Phosphonium ylide MIRC reactions exist since 1988 (Krief et al. DOI)
First of is Aggarwal whose group in 2001 building on asymmetric epoxidations and aziridations ( DOI) invented an complex indirect sulfonium ylide method (DOI).
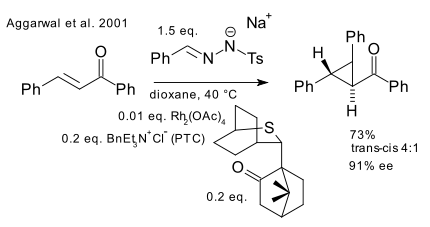
The main substrate is chalcone reacting with a chiral sulfonium ylide derived from a chiral sulfide and the carbene Rh=CHPh (generated in-situ from a tosyl hydrazone and rhodium(II) acetate). The 2001 report has gathered 114 citations to date but the cyclopropanation method described has not yet been field-tested.
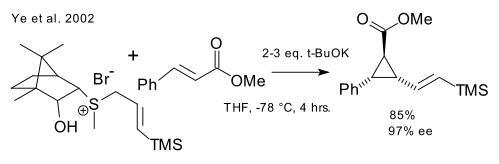
A direct chiral sulfonium ylide method is reported by Tang (Ye et al. 2002, 46 citations DOI).
Next up is Gaunt et al. (the Stephen Ley laboratory) who in 2004 came with the idea to employ ammonium ylides as Michael donors with a cinchona alkaloid as chiral catalyst. (DOI).
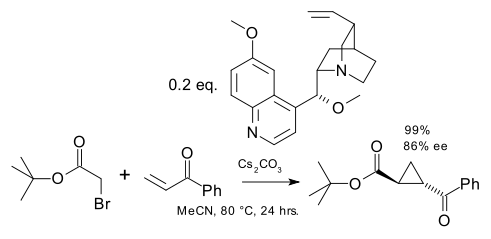
This reaction is organocatalytic. In the reaction mechanism the tertiary amine displaces a halide group in a haloketone and then a base (caesium carbonate) abstracts a proton to the ammonium ylide.
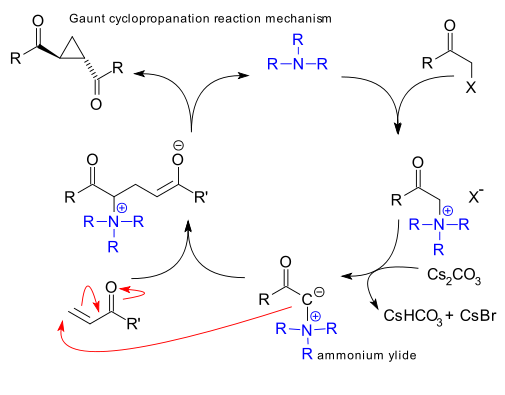
The first report clocks around 50 citations to date but is applied only once in a total synthesis of eicosanoids(DOI) and not very successfully as enantioselectivity for the particular step is poor.
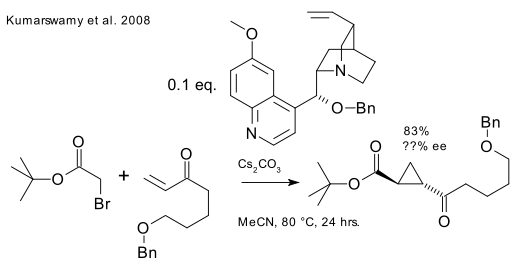
A third method (Kunz/MacMillan, 2005) is a direct sulfonium ylide method DOI. An improved catalyst exists (DOI).
Other ylide reactions are reported: those base on arsenic (Zhao et al. 2007 DOI), those based on tellurium (Liao 2003 DOI) and those with azetidinium ylides (Couty 2007 DOI)
