The OCZnNNa cycle
05 November 2009 - Bimetallic bases
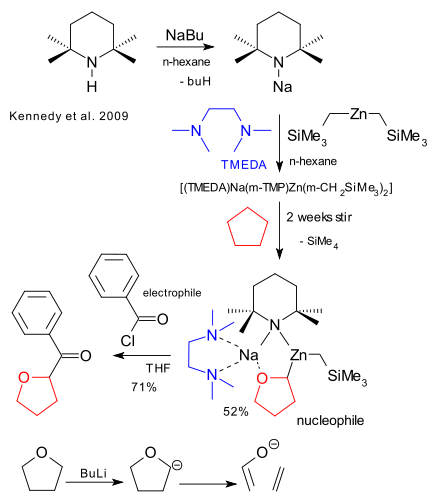
Breaking news in the field of chemistry dealing with otherwise unbreakable carbon-hydrogen bonds. The reaction: C-H activation, the substrate tetrahydrofuran (THF), the reagent: a so-called bimetallic base. Ordinary bases such as buLi can metallate the alpha hydrogen atom in THF but with subsequent ring-cleavage. Kennedy et al. now demonstrate that by adding a second metal to lithium and something else, lithiated THF can be stabilized or rather sedated (see title) (DOI).
Nitrogen cycle TMP is reacted with organosodium compound NaBu to TMPNa. The organozinc compound Zn(CH2SiMe3)2 is added next together with metal ligand TMEDA forming the actual reagent TMEDA(TMP)ZnR2. The molecular structure of the THF adduct contains a O-C-Zn-N-Na- cycle that looks odd and esoteric but makes sense. THF is stabilized by two metal chelating interactions (Zn-C and Na-O) and both metals in turn are stabilized by three nitrogen atoms. The complex is found to react predictably with common electrophile benzoyl chloride.
More bimetallic bases (Knochel base) in Grignards on lithium
Kennedy, A., Klett, J., Mulvey, R., & Wright, D. (2009). Synergic Sedation of Sensitive Anions: Alkali-Mediated Zincation of Cyclic Ethers and Ethene Science, 326 (5953), 706-708 DOI: 10.1126/science.1178165
