Perylene synthesis makeover
04 November 2010 - Organic chemistry
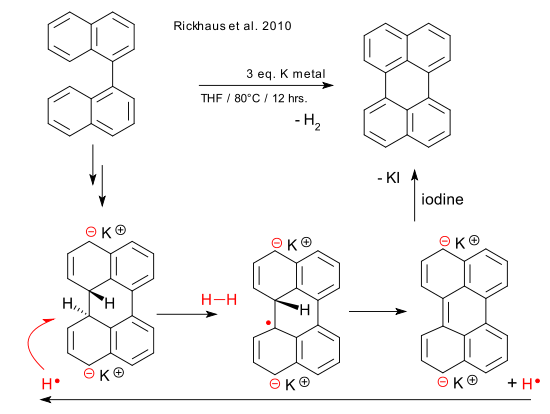 In organic reaction makeovers, ugly and badly performing reactions are metamorphosed into clean happy synthetic procedures with general utility. A recent ugly duckling getting the makeover is the conversion of 1,1-binaphthyl to perylene using an alkali metal as reported by Rickhaus et al (DOI). This variation of the Scholl reaction was discovered by Solodovnikov in 1967 and quickly forgotten about because the publication was in Russian. The authors complain that they had to do the translation themselves because of the poor Chemical Abstracts summary which makes you wonder exactly how much decent Soviet-era chemical research is hidden away. In the new and improved synthesis (quantitative yield!) the solvent dimethoxyethane was replaced by tetrahydrofuran with an optimum 3 equivalents of potassium metal. The temperature was increased from room temperature to 80°C. As always the devil is in the details: note 20 specifies that "Not cleanly cutting all sides of the potassium piece generally leads to a significant drop in yield."
In organic reaction makeovers, ugly and badly performing reactions are metamorphosed into clean happy synthetic procedures with general utility. A recent ugly duckling getting the makeover is the conversion of 1,1-binaphthyl to perylene using an alkali metal as reported by Rickhaus et al (DOI). This variation of the Scholl reaction was discovered by Solodovnikov in 1967 and quickly forgotten about because the publication was in Russian. The authors complain that they had to do the translation themselves because of the poor Chemical Abstracts summary which makes you wonder exactly how much decent Soviet-era chemical research is hidden away. In the new and improved synthesis (quantitative yield!) the solvent dimethoxyethane was replaced by tetrahydrofuran with an optimum 3 equivalents of potassium metal. The temperature was increased from room temperature to 80°C. As always the devil is in the details: note 20 specifies that "Not cleanly cutting all sides of the potassium piece generally leads to a significant drop in yield."
That leaves the reaction mechanism. Curiously in this reaction and eagerly pointed out by the authors, both binaphthyl and potassium are being oxidized, so what exactly is the reduced product? A clue was already provided by Solodovnikov: he detected hydrogen gas. In a new proposal the biphenyl is first reduced to the radical anion (as in a Birch reduction) and then again to the di-anion and at some point a new carbon carbon bond is formed. Related research had already established that the hydrogen atoms in the new bond have a trans relationship and therefore direct formation of hydrogen is unlikely. More plausible is the involvement of a free radical reaction with a hydrogen radical abstracting one of the hydrogen atoms to form H-H and then rearomatisation and expulsion of a new hydrogen radical. The solvent THF is also a radical acceptor but merely shuttles the electrons around.
