NNNS Chemistry blog
Prevous: Retire hybrid atomic orbitals? Not yet!
Next: Chandross on organic solar cells
Methylhydroxycarbene
26 June 2011 - Quantum mechanics
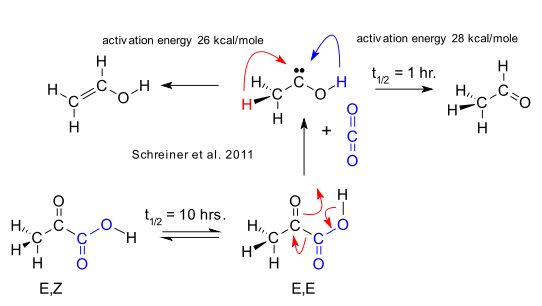 After hydroxymethylene (2008) and phenylhydoxymethylene (2010, DOI), Peter Schreiner and group now present methylhydroxycarbene. The method of generation is roughly similar: heat a precursor molecule to really high temperatures in Flash vacuum thermolysis and then trap the reaction products in solid argon at really low temperatures. In the case of methylhydroxycarbene the variables are pyruvic acid, 900°C and 11 K. The carbene yield is 2 to 5%. When irradiated at 435 nm, the carbene rearranges in a hydrogen 1,2-rearrangement to vinyl alcohol and acetaldehyde as can be observed by infrared spectroscopy. Acetaldehyde is formed predominantly with a chemical half-life of 1 hour. This is fast compared to a half-life of 10 hours for the isomerization of oxygen-hydrogen bond in pyruvic acid. The computed activation energies on the other hand are very high: 28 kcal/mole for the transition to acetaldehyde and 26 kcal/mole for the one to vinyl alcohol. If this reaction was simply ruled by Thermodynamic versus kinetic reaction control with kinetic control prevailing the reaction product would be vinyl alcohol and not acetaldehyde.
After hydroxymethylene (2008) and phenylhydoxymethylene (2010, DOI), Peter Schreiner and group now present methylhydroxycarbene. The method of generation is roughly similar: heat a precursor molecule to really high temperatures in Flash vacuum thermolysis and then trap the reaction products in solid argon at really low temperatures. In the case of methylhydroxycarbene the variables are pyruvic acid, 900°C and 11 K. The carbene yield is 2 to 5%. When irradiated at 435 nm, the carbene rearranges in a hydrogen 1,2-rearrangement to vinyl alcohol and acetaldehyde as can be observed by infrared spectroscopy. Acetaldehyde is formed predominantly with a chemical half-life of 1 hour. This is fast compared to a half-life of 10 hours for the isomerization of oxygen-hydrogen bond in pyruvic acid. The computed activation energies on the other hand are very high: 28 kcal/mole for the transition to acetaldehyde and 26 kcal/mole for the one to vinyl alcohol. If this reaction was simply ruled by Thermodynamic versus kinetic reaction control with kinetic control prevailing the reaction product would be vinyl alcohol and not acetaldehyde.
According to Schreiner the reaction proves that besides thermodynamic / kinetic control we can now add a third reaction driver: quantum tunneling It is the same tunneling also appearing in the kinetic isotope effect and it means that a reaction along a reaction coordinate does not bother to scale the barrier height but simple tunnels its way through the barrier itself. In this event the barrier width takes on some significance and as it happens this width is relatively narrow in the conversion to acetaldehyde.
