Asymmetric alpha C-H activation
25 September 2016 - Organic reactions
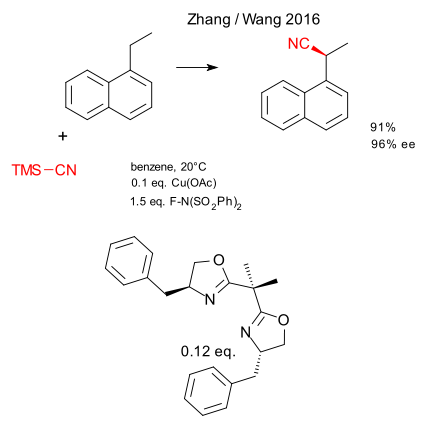 A recent contribution from the Guosheng Liu laboratory concerns the asymmtric version of a copper catalysed alpha C-H-activation (Wen Zhang , Fei Wang DOI). This work builds on the classic Kharasch-Sosnovsky reaction.
A recent contribution from the Guosheng Liu laboratory concerns the asymmtric version of a copper catalysed alpha C-H-activation (Wen Zhang , Fei Wang DOI). This work builds on the classic Kharasch-Sosnovsky reaction.
The substrates are benzylic compounds with unactivated protons at the alpha position, for example 1-ethylnaphthylene. The oxidant N-fluorobenzenesulfonylimide generates the achiral free radical . This cyanation proceeds with trimethylsilyl cyanide. The catalyst system is copper acetate and a custom-built bulky chiral bisoxazoline ligand. In the transition state copper has two cyano groups and is coordinated to both ligand nitrogen atoms with the remaining coordination site interacting with the alpha methylene group. Yield 91%, enantiomeric excess 96%.
