NNNS Chemistry blog
Prevous: The cathones have it, the cathones have it!
Next: Penta aqua nitrosyl iron sulphate
Ozonolysis latest
19 May 2019 - Organic chemistry
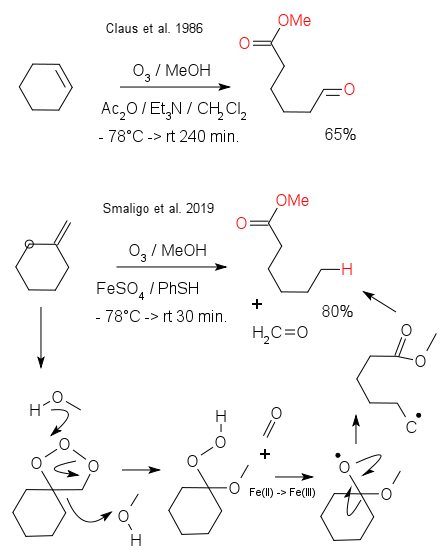 As pointed out by Smaligo et al. in a recent article in Science,(DOI) in organic synthesis it is not always about combining small molecules to create larger ones but occasionally it can be about cutting existing bonds. Ozonolysis is a classic reaction involving the cleavage of an alkenes, for example that of cyclohexene to methyl-6-oxo-hexanoate. (DOI) In cyclohexene both carbon atoms on the chopping board have sp2 hybridisation but as an expansion of the available toolbox the Smaligo team has have come up with hydrodealkylenylation with an sp3 - sp2 carbon bond. The name is a play on hydrodealkylation with for example the conversion of toluene to benzene.
As pointed out by Smaligo et al. in a recent article in Science,(DOI) in organic synthesis it is not always about combining small molecules to create larger ones but occasionally it can be about cutting existing bonds. Ozonolysis is a classic reaction involving the cleavage of an alkenes, for example that of cyclohexene to methyl-6-oxo-hexanoate. (DOI) In cyclohexene both carbon atoms on the chopping board have sp2 hybridisation but as an expansion of the available toolbox the Smaligo team has have come up with hydrodealkylenylation with an sp3 - sp2 carbon bond. The name is a play on hydrodealkylation with for example the conversion of toluene to benzene.
That iron could be a useful catalyst in a hydrodealkylenylation came about after digging up an old article from F.F. Rust - what is in a name - (DOI) about the conversion of hydroperoxides (an intermediate in ozonolysis) to oxyradicals in the presence of Fe(II). The final result in the new ozonolysis modification is a reagent combination of iron(II) sulfate and benzenethiol (as proton donor). In the unauthorized reaction mechanism depicted, the first stage is the molozonide, then formaldehyde is ejected, followed by reduction of OOH to the oxo radical, followed by the actual C-C cleavage reaction and with finally phenylthiol jumping in to arrest the radical. Complaints are welcome at Twitter.
Rik
