Happy birthday fullerene
04 September 2010 - Circumstellar chemistry
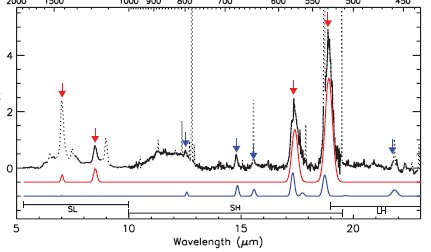 This blog would have missed the 25th birthday of the discovery of fullerene if it was not for Google who on their website replaced the second letter O by (surprise) a rotating buckyball. Fortunately for this occasion it was not difficult to find new fullerene research to report on. This week for example Cami et al. explain in the journal Science how to spot fullerenes in outer space (DOI). They pointed the Spitzer Space Telescope at one of 3000 known planetary nebula which have nothing to do with planets but everything to do with clusters of dust ejected by stars at the end of their life cycle. In one particular young system (Tc 1 if you have to know) they detected both C60 and C70 buckyballs in the produced IRS spectra.
This blog would have missed the 25th birthday of the discovery of fullerene if it was not for Google who on their website replaced the second letter O by (surprise) a rotating buckyball. Fortunately for this occasion it was not difficult to find new fullerene research to report on. This week for example Cami et al. explain in the journal Science how to spot fullerenes in outer space (DOI). They pointed the Spitzer Space Telescope at one of 3000 known planetary nebula which have nothing to do with planets but everything to do with clusters of dust ejected by stars at the end of their life cycle. In one particular young system (Tc 1 if you have to know) they detected both C60 and C70 buckyballs in the produced IRS spectra.
The region is characterised by the absence of typical hydrogen-containing molecules such as ethylene or HCN, the fullerene band-widths reveal an excitation temperature of 180 K, the fullerene molecules are expected to be neutral as typical ion peaks are absent in the spectra and the molecule is alo expected to reside on carbonaceous grain particles. The C60 molecules are hotter than the C70 molecules and therefore expected to be located on the grain closer to the central star.
The abundance of C60 in this region of space is explained by the hydrogen-poor environment. In an estimate fullerenes lock down 1.5% of all carbon. In terrestrial laboratories absence of hydrogen is also key in fullerene recipes and maximum yield also never exceeds 1.5%. As to the why the hydrogen is missing, another simple explanation: the star in question ejected its hydrogen envelope several thousands of years ago exposing the helium core that in a second thermal pulse now ejects carbon rich material as the main helium fusion product.
