Artificial leaf chemistry
04 April 2011 - Catalysis
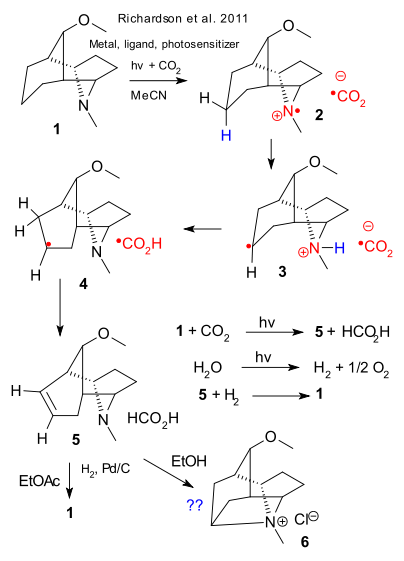 In photosynthesis water and carbon dioxide in the presence of light react together to form carbohydrates and oxygen. In artificial photosynthesis this process is recreated in the laboratory. In a recent venture Barry Carpenter has plotted the next step in photosynthesis based on a certain carbon dioxide reduction using amines (Richardson et al. 2011 DOI). In this system CO2 is reduced by a metal porphyrin or metal corrole with p-terphenyl (PTP) as photosensitiser. The metal is reactivated by an tertiary amine as sacrifical catalyst (a.k.a. reductive quencher) This reaction is well known but thus far not very practical because it has not been possible yet to generate the amine.
In photosynthesis water and carbon dioxide in the presence of light react together to form carbohydrates and oxygen. In artificial photosynthesis this process is recreated in the laboratory. In a recent venture Barry Carpenter has plotted the next step in photosynthesis based on a certain carbon dioxide reduction using amines (Richardson et al. 2011 DOI). In this system CO2 is reduced by a metal porphyrin or metal corrole with p-terphenyl (PTP) as photosensitiser. The metal is reactivated by an tertiary amine as sacrifical catalyst (a.k.a. reductive quencher) This reaction is well known but thus far not very practical because it has not been possible yet to generate the amine.
In Carpenter's novel system the amine is regenerated in a kind of internal transfer hydrogenation. The catalyst (1) reacts with CO2 to form a radical cation 2 and a CO2 radical anion. A proton is then transfered to nitrogen from the neighboring bridge and the molecules cage structure makes sure this is an efficient step. The CO2 radical anion collects two protons first from the amino group and then from the alkyl bridge in a Hofmann elimination.
The spent amino catalyst 5 now has an alkene group. In the grand scheme yet to be worked out CO2 reduction is coupled with photochemical water oxidation forming oxygen and hydrogen. Hydrogen then regenerates the catalyst. For now the Carpenter group made sure formic acid was formed and that the proposed structure for 5 was correct. Not all went according to plan: hydrogenation of 5 back to 1 with H2/Pd/C was possible in ethyl acetate but not in ethanol. The formation of 6 (after chloroform quench) is unexplained but amines are well known to behave oddly.
